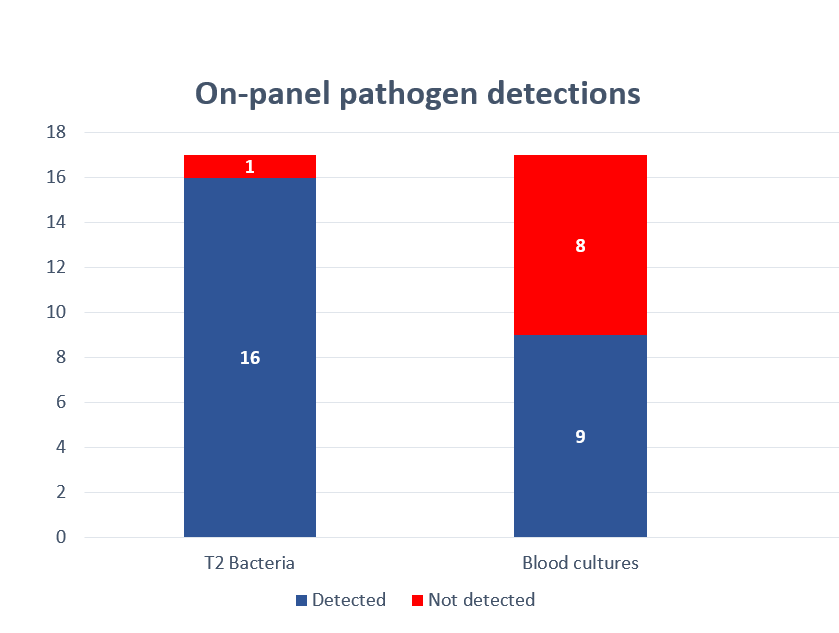
T2 Biosystems Announces FDA 510(k) Submission to Expand the Pathogen Detection on its FDA-Cleared T2Bacteria Panel to Include Detection of Acinetobacter baumannii
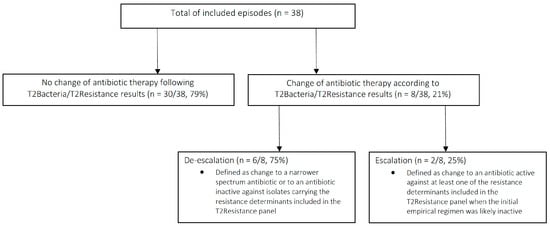
Antibiotics | Free Full-Text | T2Bacteria and T2Resistance Assays in Critically Ill Patients with Sepsis or Septic Shock: A Descriptive Experience
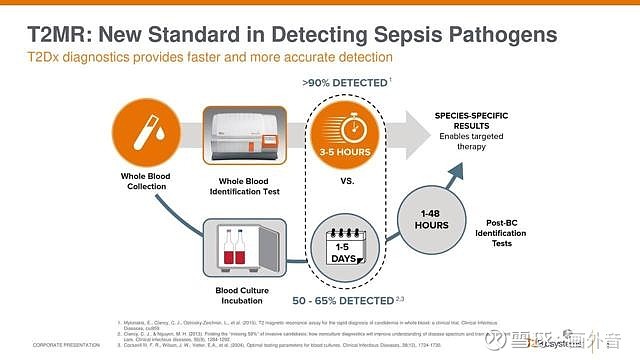
FDA's Approval of T2 Biosystems T2Bacteria Panel Underlines Importance of Sepsis Diagnosis and Treatment | BioSpace

A culture-free biphasic approach for sensitive and rapid detection of pathogens in dried whole-blood matrix | PNAS

Direct detection of ESKAPEc pathogens from whole blood with the use of the T2Bacteria Panel allows early antimicrobial stewardship intervention in patients with sepsis

Effective Rapid Diagnosis of Bacterial and Fungal Bloodstream Infections by T2 Magnetic Resonance Technology in the Pediatric Population | Journal of Clinical Microbiology